Written by: Sarah W. Denton, Research Fellow, Institute for Philosophy and Public Policy, George Mason University; Research Assistant, Science and Technology Innovation Program (STIP), The Woodrow Wilson International Center for Scholars
On June 19, 2018, the National Academies of Sciences, Engineering, and Medicine (NASEM) held a public briefing timed with the release of a new report titled, Biodefense in the Age of Synthetic Biology. The panel – comprised of the committee chair, Dr. Michael Imperiale, and four authors of the report, Dr. Patrick Boyle, Dr. Peter Carr, Dr. Diane DiEuliis, and Dr. Jill Taylor– gathered to present their research to the public and provide an opportunity for an in-person and online question and answer session.
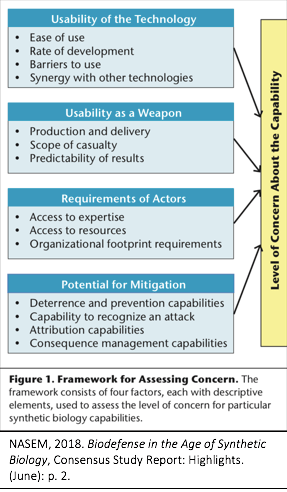 During this briefing, the panel discussed the study’s concern-assessment framework (Figure 1). The framework consists of four factors that can be used to assess the level of concern for current and future synthetic biology capabilities. Notably, rather than attempting to assess the levels of concern presented by various technologies (e.g., CRISPR/Cas9), this framework focuses instead on assessing capabilities “that potentially pose a concern because of the harm they might enable.”[i]
During this briefing, the panel discussed the study’s concern-assessment framework (Figure 1). The framework consists of four factors that can be used to assess the level of concern for current and future synthetic biology capabilities. Notably, rather than attempting to assess the levels of concern presented by various technologies (e.g., CRISPR/Cas9), this framework focuses instead on assessing capabilities “that potentially pose a concern because of the harm they might enable.”[i]
While the framework draws on previous works (e.g., Tucker 2012and the 2004 Fink report), what makes this report unique is its use of the Design-Build-Test (DBT) process as the foundation for its capability-assessment. DBT is the “iterative process of designing a prototype, building a physical instantiation, testing the functionality of the design, learning from its flaws, and feeding that information back into the creation of a new, improved design.”[ii]Specifically, the committee found it useful to conceptualize current and future technological developments in terms of the ways in which they enable the DBT cycle, granting that it is entirely possible for some technologies to have impacts across multiple phases of the DBT cycle.[iii]For example, the committee identified potential points of concern in all phases of the DBT process in their analysis of the level of concern relating to the re-creation of known pathogens.[iv] Continue reading “NASEM Report: Biodefense in the Age of Synthetic Biology”

 In late June, I attended the
In late June, I attended the  To begin, Dr. Millet, Vice President of Safety and Security at iGEM, described the system put in place at iGEM to manage and mitigate biosafety and biosecurity risks. The iGEM competition is an international synthetic biology competition in which teams of students use standard biological parts to genetically engineer organisms to address real world problems. Last year, approximately 6,000 participants from 45 countries were part of the iGEM competition. At iGEM, everyone is responsible for safety, so teams, supervisors, and principal investigators are expected to take an active role. Resources on the iGEM
To begin, Dr. Millet, Vice President of Safety and Security at iGEM, described the system put in place at iGEM to manage and mitigate biosafety and biosecurity risks. The iGEM competition is an international synthetic biology competition in which teams of students use standard biological parts to genetically engineer organisms to address real world problems. Last year, approximately 6,000 participants from 45 countries were part of the iGEM competition. At iGEM, everyone is responsible for safety, so teams, supervisors, and principal investigators are expected to take an active role. Resources on the iGEM 