Happy New Year from the Pandora Report! We hope everyone had a jolly and healthy holiday season! January is One Health Awareness Month to promote the approach of greater cross-sectoral collaboration and communication across the human-animal-environment interface. In big news, Danyale C. Kellogg, a Biodefense PhD student, is taking over as the Managing Editor of the Pandora Report and we cannot wait to see more of her insights and expertise.
Remembering Stephanie Smith
The Biodefense Graduate Program is sad to report the recent passing of alumna Stephanie Smith (MS ’17). Stephanie had a love of science, boundless curiosity, a keen mind, and engaging personality. She made the most of her time in the program and in her role as the Science and Technical Advisor to the US Postal Inspection Service she exemplified the Biodefense program’s ideal of bridging the gap between science and policy. The Biodefense Program offers her family and friends our deepest condolences.
January Is One Health Awareness Month!
To promote collaboration between animal, environmental, plant and public health scientists, on December 19, 2019 the US Senate unanimously passed a bipartisan Senate Resolution 462 (S Res 462) introduced by Senators Dianne Feinstein (Democrat-California) and Martha McSally (Republican-Arizona) designating January 2020 as “National One Health Awareness Month” in the US. Excitingly, the campaign was picked up and promoted by One Health advocates around the world. Help keep the momentum going! To help spread the word, you can use the hashtags #OneHealth and #OneHealthAwarenessMonth!
3 Issues to Watch in Global Health in 2022
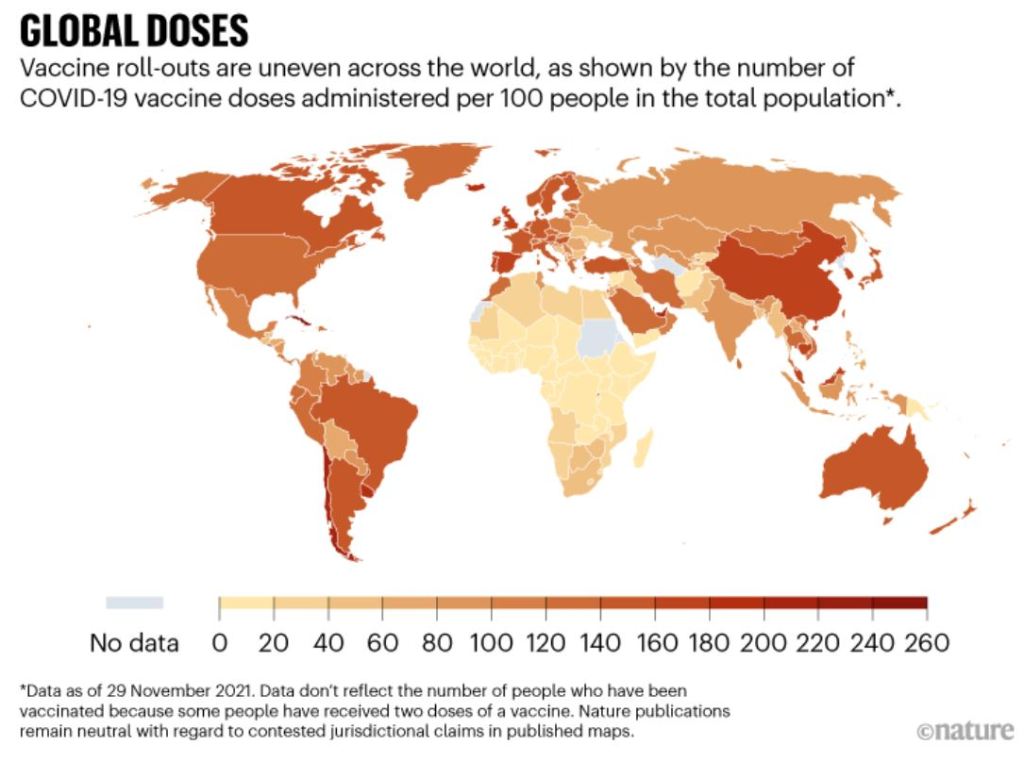
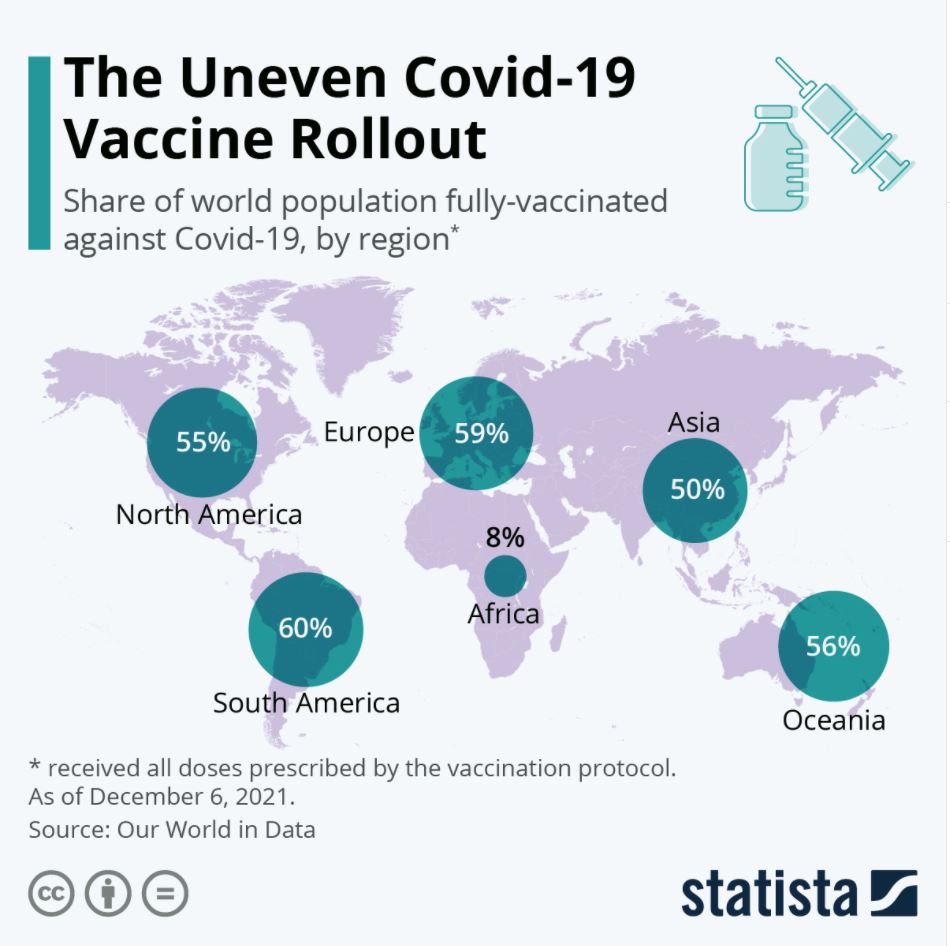
As we enter the third year of COVID-19, STAT has identified three issues in the world of global health and infectious diseases that are worth keeping an eye on. (1) Will 2022 be the year Covid starts to settle down, to show signs of being more endemic, less pandemic? This year, COVID-19 vaccine doses should become more widely used in lower income nations. But, will these vaccines need to be updated to better combat the emerging variants? (2) What will happen with the World Health Organization (WHO) this year? The WHO has come under much scrutiny the last couple years, and funding has been a struggle. Also, the World Health Assembly will soon vote on whether to give Tedros another five years at the helm. (3) How will vaccination rates fair in 2022? Lower income countries have struggled to get their hands on enough doses, but that will hopefully change in 2022. On the other hand, the US has seen great opposition to COVID-19 vaccine mandates and many are still unvaccinated.
10 Key Global Health Moments From 2021
The World Health Organization (WHO) shared 10 global highlights from 2021 ranging from innovation in vaccines to inequities in vaccine distribution, but also a new malaria vaccine and a drop in tobacco use. The COVID-19 pandemic has continued to strain the health systems of nations in conflict, such as Yemen and Syria. The WHO states that the pandemic is “likely to halt two decades of global progress towards universal health coverage (UHC), having triggered the worst economic crisis since the 1930s and badly disrupted health services.” Health systems overcoming the challenges of 2021 are doing so in large part thanks to the “huge sacrifices of the women who represent 70% of the health and social workforce.” 2021 marked the 100th anniversary of the discovery of insulin and the WHO launched a Global Diabetes Compact to “speed up action on the prevention and treatment of diabetes.” Read more here.
Multi-Service Tactics, Techniques, and Procedures for Domestic Chemical, Biological, Radiological, and Nuclear Response
This publication provides doctrinal guidance for executing domestic chemical, biological, radiological, and nuclear CBRN response operations under the auspices of defense support of civil authorities (DSCA) and describes Department of Defense (DOD) roles and responsibilities in conducting domestic CBRN responses. It also describes the framework for domestic response, operational fundamentals, and DSCA.
Chapters are oriented around DOD support for domestic CBRN incidents as a component of the National Response Framework (NRF) to provide organizational understanding and knowledge of response activities in support of a domestic CBRN response. The publication contains appendixes that present additional details on specific domestic CBRN response organizations and their associated capabilities and selected related topics. This publication reflects changes in the capabilities within the CBRN response enterprise and recent changes in associated guidance, methodology, and terminology.
Chapter 1 provides an overview of the national frameworks for US domestic response. Chapter 2 focuses on initial planning and anticipation of CBRN incident response and activities, in line with the CBRN response phases of shape and anticipate. Chapter 3 focuses on the phases of respond and operate. Chapter 4 focuses on the phases of stabilize and transition. Read the publication here.
CBRN Terrorism Interdictions (1990–2016) and Areas for Future Inquiry
The pursuit and use of chemical, biological, radiological, and nuclear (CBRN) weapons has been examined by scholars for more than two decades. What has not been examined are the cases in which non-state actors were prevented from obtaining or using these weapons and agents and the corresponding reasons for successful interdiction. This article uses the Profiles of Incidents Involving CBRN and Non-State Actors (POICN) database to carry out an exploratory analysis of CBRN interdictions around the world from 1990–2016. Using basic descriptives and cross-tabulations, this study finds that successful interdictions often resulted from probable cause searches, surveillance operations, but also from other, unknown reasons. However, there is a tremendous amount of variation when it comes to modes of interdiction and actor motivation. The same goes for jurisdiction, whether international collaboration aided the interdiction, and weapon acquisition and delivery. This text is intended to serve as a foundation for the study of CBRN terrorism interdiction as it seeks to undercover why some law enforcement efforts fail while others succeed. The author, Dr. Wesley D. McCann, is an Assistant Professor in the Department of Criminology, Law and Society at GMU. Read the article here.
A Handbook for Ending Catastrophic Biological Risks: How the United States Can Prevent Future Pandemics and Deter Biological Weapons
In a time of daily confusion in the public discourse on the current pandemic, the Janne E. Nolan Center on Strategic Weapons, an institute of the Council on Strategic Risks (CSR), is releasing A Handbook for Ending Catastrophic Biological Risks: How the United States Can Prevent Future Pandemics and Deter Biological Weapons. Authored by some of the nation’s top experts in combating biological risks, this unique guide presents a “clear, viable and improved path” toward addressing them. It calls for a US government surge of investments in addressing biological threats of all origins, with detailed recommendations for improving US government programs and cooperative partnerships.
The Handbook for Ending Catastrophic Biological Risks starts with a bold, moonshot-level vision that can drive transformative changes—not the incremental improvements the US government has often pursued in the past. That vision has two critical parts:
The Handbook promotes an ideal yet achievable vision on naturally-arising biological risks: preventing future pandemics – moving beyond simply enduring them and accepting the lives lost and damage to health security, national security, and the economy that pandemics can cause.
The Handbook also details a complementary vision regarding deliberate biological risks (one which will require far more significant shifts in the nation’s goals and policies), asserting that: The United States should lead the world in making biological weapons the first category of weapons of mass destruction to be effectively eliminated or rendered obsolete. To advance this vision, the report recommends that national security leaders adopt a deterrence by denial strategy that focuses on denying an attacker success in their likely aims regarding biological weapons, such as causing mass casualties, mass confusion, and erosion of operational capabilities.
Dr. Yong-Bee Lim, recent graduate of the Biodefense PhD program, is among the authors of The Handbook. Find The Handbook here.
Second WHO Consultative Meeting on a Global Guidance Framework to Harness the Responsible Use of Life Sciences
On 7 September 2021, 47 participants attended the second World Health Organization (WHO) consultative meeting on the development of the global guidance framework to harness the responsible use of life sciences. Participants were drawn from all six WHO regions and brought a wide range of expertise, representing academia, scientists and researchers, publishers and editors, public health officers, policymakers, research donors, and representatives from regional and United Nations entities.
The objectives of the meeting were to discuss the findings and recommendations of the three working groups, which were established as a follow-up activity of the first consultative meeting held on 11 March 2011. In addition, participants were invited to discuss the next steps in the development of the global guidance framework to harness the responsible use of life sciences (the Framework). Finally, this meeting was intended to pursue the consultation and engagement processes on this area of work.
Participants welcomed and supported the findings and recommendations of the three working groups. The set of guiding values and principles underpinning the Framework was adopted and common understanding was reached on tools and mechanisms for governance and awareness raising, education, capacity building and engagement activities. It was agreed to link and cross reference the three documents developed by the working groups, noting the need for consistent language and terminologies across the three documents. The comprehensiveness of these documents was highlighted along with the need to have a practical and actionable Framework, which would refer to the current working groups’ themes.
Future activities include the development a glossary of key terminologies and the work on a series of case studies and scenarios to test the utility of the Framework and to demonstrate how the different elements of the Framework could be used in real and hypothetical situations. Two working groups will be established to carry out these activities. WHO will continue to consult with relevant individuals, groups, Member States and the regions on the development, dissemination and implementation of the Framework in the coming months. Read the report here.
ALL THINGS COVID-19
The Latest on Omicron
The Omicron variant of SARS-CoV-2 is spreading more easily than the original virus, but it “likely carries a lower chance of getting seriously ill.” There is preliminary evidence that the “risk of being admitted to the hospital or the intensive care unit during the omicron surge in the US is about half of the risk observed during the delta surge.” According to Dr. Pamela Davis, who’s a pulmonologist at Case Western Reserve University, “in the older age group, it’s still a nasty disease, even if it’s less [nasty] than the delta variant.” Now, in the US, there are 126,000 hospitalizations and more than a quarter of ICU beds are filled by COVID-19 patients. A recent study showed that vaccination reduces a person’s risk of severe illness and hospitalization.
Dr. Eric Topol provides an easy-to-follow overview of the Omicron variant of COVID-19, including how the vaccines combat this variant. Topol states, “while we have a very smart immune system that is vaccine- trained to defend surprisingly well against severe disease by a hyper-mutated, hyper -transmissible virus, too many humans have not gotten on board. We’ll nevertheless win this battle, fortunate to have 2 major points in our favor.”
700 Sheep and Goats Were Arranged in the Shape of a Syringe to Encourage Vaccinations
South of Hamburg, Germany, 700 sheep and goats were arranged into the shape of a 330-foot syringe. Shepherd Wiebke Schmidt-Kochan spent several days practicing with her animals to create the syringe as encouragement for people who are still hesitating to get vaccinated. In Germany, 71.2% of the population has received at least two shots and 38.9% has received a booster shot.

The US COVID-19 Vaccination Program at One Year: How Many Deaths and Hospitalizations Were Averted?
Nearly 800,000 Americans have died so far during the US COVID-19 pandemic, with more than half those deaths occurring during 2021. One year into the US vaccination effort, much attention has focused on the stubborn persistence of the pandemic, which has been fueled by new, more- transmissible variants and the millions of Americans who have not gotten their shots. However, the positive impact of the rapid development and deployment of highly effective vaccines — the reduction in deaths and hospitalizations — has been less obvious.
In July, The Commonwealth Fund reported that the US vaccination program had averted 279,000 deaths and 1.25 million hospitalizations, primarily by blunting a surge in the Alpha variant during spring 2021. Since that report, nearly all of the US has experienced a wave of infections, hospitalizations, and deaths caused by the highly transmissible Delta variant. More than 1,000 Americans are dying each day.
In an updated report, The Commonwealth Fund updates its estimates, through the end of November 2021, of COVID-related deaths and hospitalizations avoided because of the US vaccination program. Briefly, the agent-based computer model analyzes features of the coronavirus, its transmission, and its effects to compare the observed pandemic trajectory (infections, hospitalizations, and deaths) to a counterfactual scenario in which no vaccination program exists. The model incorporates the transmission dynamics of previous variants other than Omicron, which is only now beginning to appear in the US. The model accounts for waning immunity and changes in population behavior over time as schools and businesses have reopened and travel has increased.
In the absence of a vaccination program, there would have been approximately 1.1 million additional COVID-19 deaths and more than 10.3 million additional COVID-19 hospitalizations in the US by November 2021. Without the US vaccination program, COVID-19 deaths would have been approximately 3.2 times higher and COVID-19 hospitalizations approximately 4.9 times higher than the actual toll during 2021. If no one had been vaccinated, daily deaths from COVID-19 could have jumped to as high as 21,000 per day — nearly 5.2 times the level of the record peak of more than 4,000 deaths per day recorded in January 2021. Read the full report here.
17 Pandemic Innovations That Are Here to Stay
POLITICO asked its reporters to “identify the pandemic-induced policy innovations that have changed life in America over the last year and a half.” Following are a handful of the ways that the pandemic “spurred innovation in America, mostly for the better”:
- Cocktails-to-go
- Telehealth
- mRNA vaccines
- Robot deliveries
- “Warp speed” government investing
- “Streateries”
10 Lessons I’ve Learned From the COVID-19 Pandemic
STAT writer Helen Branswell looks back on the COVID-19 pandemic thus far and highlights 10 lessons learned from the last two years. The first is the “you gotta act fast,” especially in an outbreak of a novel pathogen. In early 2020, the world had hesitated too long and lost its chance to quickly contain the virus. Another hard lesson is that “even in the face of a deadly pandemic, politics override public health.” The pandemic showed that “political leaders [will] put the lives of their citizens at risk by downplaying or downright lying about a disease outbreak, just because telling the truth might jeopardize their political fortunes.” Much of the US response to COVID-19 has been “broken down along political lines.” This is exemplified by the lower vaccine rates red counties versus blue. Read more about COVID-19 lessons learned here.
Department of Defense Contributions to the US COVID-19 Response, at Home and Abroad
The US Department of Defense (DOD) should be systematically incorporated into any US government vision on international global health security. The United States should build on what has been learned and achieved through DOD support at home to the civilian-led COVID-19 response, as well as past DOD contributions to pandemic response overseas, and incorporate long-standing DOD international capabilities against biological threats. DOD should contribute to responding to the acute phase of the pandemic and build long-term preparedness capacities.
DOD has considerable assets in three areas that can and should be included, when appropriate, to support the US civilian-led international response:
- Logistics, lift, and planning;
- Biosurveillance and infectious disease research and development; and
- Relationships built through decades of international collaboration in biosecurity and biosafety, ongoing global health engagements, and work in overseas laboratories.
DOD already is playing a significant role in the procurement and delivery of 1 billion doses of the Pfizer-BioNTech vaccine, which President Biden has pledged to the COVAX vaccine solidarity mechanism to benefit 92 low- and lower-middle-income countries. A process of strategic planning for other likely DOD contributions to contain the global COVID-19 pandemic should begin right away. Read the full commentary from the Center for Strategic and International Studies (CSIS) here.
COVID Helped Cause the Biggest Drop in US Life Expectancy Since WWII
The pandemic has helped take 1.8 years off the average life expectancy for Americans in 2020, based on the latest federal mortality data. This marks the greatest change in the average American lifespan since World War II. In 2020, COVID-19 was the “third-most common cause of death in the United States, with one out of 10 fatalities due to the virus.” Dr. Zinzi Diana Bailey, a social epidemiologist at the University of Miami’s Miller School of Medicine, said, “A loss of two years seems limited, but that’s rolling back decades and decades of progress.” Death rates increased for all people aged 15 years or older in 2020. Though people ages 85 years or more died more frequently than the other age groups, people aged 35-44 years saw the biggest increase in death rates. One spot of good news is that the national infant mortality rate dropped 2.9%, a record low according to the Centers for Disease Control and Prevention.
EVENTS
Online International Symposium: COVID-19 Under the Pandemic Mass Gathering Events and Public Health Measures
When holding a mass gathering event represented by a large-scale international event such as the Olympic Games, it is necessary to prepare and respond to various risks. Japan has experienced many large international mass gathering events. In Japan, the focus has been on securing emergency medical care and mass disaster systems for such events, but in recent years there has been increasing interest in public health crisis management systems for infectious diseases and the like. The outbreak of the SARS-CoV-2 pandemic provided an opportunity to draw attention to the public health crisis management system in mass gathering. In addition to ensuring the safety of event participants, mass gathering is also required to be prepared to have a negative impact on the local health and public health system. The purpose of this symposium is to look back on the measures against mass gathering in the pandemic disaster and to obtain suggestions for future measures against mass gathering and measures against new coronavirus infectious diseases. This Zoom webinar will be held on 13 January. Register here.