Happy Friday! Do you remember the Jurassic Park character Dr. Ian Malcolm and his famous “life finds a way” quote? Well, in this case, nature is finding its own resistance against gene drive in the wild.
Bioterrorism Preparedness & Response Position Paper 
The InterAgency Board (IAB) has released their proposed model for bioterrorism response: initial operations and characterizations. “Under this model, responder organizations that meet eligibility requirements can apply to operate through contracts as approved bioterrorism response organizations within their own jurisdictions. These teams would be trained and equipped to meet a set of national standards and would work collaboratively with the Federal Bureau of Investigation (FBI) and the Centers for Disease Control and Prevention (CDC) Laboratory Response Network (LRN) in bioterrorism incident responses.” Within the report, you can find training standards and procedures for sampling and field biodetection devices. There is also a substantial section on funding that emphasizing the necessity of implementing and sustaining the bioterrorism response model via funding. The funding plan involves “three types of expenses: annual national program costs for WMD-CST and LRN participation of $22,237,824; participating response organizations start-up costs (per team) of $353,660—developed using a notional community;27 and annual participating response organizations costs of $66,332. This model does not address costs for validating field detection equipment performance, which could be significant.” This particular section breaks down costs that range from equipment maintenance to depreciation.
GMU Biodefense Master’s Open House
Looking to study about everything from anthrax to zika while advancing your education? Check out our biodefense master’s open houses – Thursday, February 16th and Wednesday, March 22nd at 6:15pm at our Arlington Campus. These open houses are a great opportunity to learn more about the GMU biodefense program, speak to a professor, and mingle with other biodefense gurus!
2017 ASM Biothreats Conference
The meeting on biothreat research, response, and policy is just around the corner and the Pandora Report is your source for this wonderful event! Registration is still open and we’ll be having four on-the-ground biodefense graduate student reporters giving us all the great updates from this three-day event. We’ll be live tweeting during the meeting and providing a substantial overview regarding certain sessions and more.
 GMU Biodefense Director Talks Growing Threats and Lack of Action
GMU Biodefense Director Talks Growing Threats and Lack of Action
Take a venture down the biodefense rabbit hole with Dr. Gregory Koblentz! A member of the Scientist Working Group on Chemical and Biological Weapons at the Center for Arms Control and Non-Proliferation, you could say that Dr. Koblentz eats, sleeps, and breathes biodefense. His most recent work has looked at the role of responsible science in biodefense programs, dual-use research of concern, and the the growing concern of biosecurity/biosafety. In a recent report, “Koblentz indicates two factors that caused a concerning increase in the number of biodefense programs worldwide. The first was the global fear of the bioterrorist threat in the aftermath of 9/11, especially after the 2001 anthrax attacks. Also, since 2003, there have been several infectious disease outbreaks with global impact that caught people by surprise: SARS, H5N1, H1N1, Ebola and Zika. ‘There’s been a growth in the number of biosafety laboratories that are safe enough to do this work on these kinds of pathogens both because they’re caused by natural causes and also because of the fear of terrorists getting a hold of them.’ The growth in the number of programs poses additional risks themselves. Even though the biodefense programs are created as a means of stopping threats, the increased number of programs means that there are more chances for something bad to happen.” Dr. Koblentz points to the changing nature of biological threats – from state bioweapon programs to non-state actors and even naturally occurring outbreaks. The most recent BWC Review Conference is also a topic of concern for Dr. Koblentz, as he notes that it was “huge missed opportunity, and will setback efforts to reduce the risks posed by biological weapons and bioterrorism. At the outset of the conference [it] looked good. But in the final days of the conference, Iran sabotaged the proceedings and blocked the consensus needed to adopt any of these measures”
U.S. Biotech Rule – A Mixed Bag of Promises and Perils 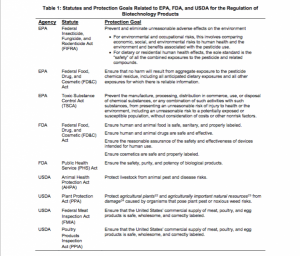
Regulations and policies have been struggling to keep up with the trajectory of genetic engineering. Reviewing these regulations falls on the FDA, EPA, and USDA, which means that they’re responsible for maintaining as modern and relevant practices as possible. Gene editing tools like CRISPR challenge these often slow efforts, however a new proposal was recently released, focusing on the path to market. This pathway, while built with good intentions, is often plagued with cracks. “Earlier this month, the White House released an update to the overarching system of biotech regulation, known as the Coordinated Framework for Regulation of Biotechnology. But it’s still up to individual agencies to clarify how they intend to classify and evaluate various GE products. In an apparent effort to get plans on the table before a change of administration, USDA and FDA put out draft proposals on 18 January addressing several categories of GE products”. As we reported a few weeks back, efforts to use genetically modified mosquitos to combat Zika, were met with residential resistance, which makes these regulations all the more sensitive. The FDA is responsible for overseeing “technologies for sterilizing and controlling animal populations, but giving it responsibility for gauging the environmental impact of a mosquito raised eyebrows on both sides of the debate”. Many have noted that the FDA truly doesn’t have the capacity to review such work in a timely manner. What about genetically engineered plants? Well, the USDA’s APHIS has specific definitions for what defines GE plants, however this definition previously focused on the production process and not the end product. “The proposed rule exempts certain products from the definition of GE, including plants containing inserted DNA from a sexually compatible species, and plants with DNA changes that could also be achieved through older chemical or radiation-based methods.” While many say that this change is good, others worry that the regulations tend to focus on projects that larger companies employ, while smaller companies lack the capacity for such controversial work, meaning that these regulations could inhibit their work.
The Cost of Cooperation in Global Health
A recent publication in The Lancet looks to the financial backing for global health cooperation via the WHO, the World Bank, the Global Fund to Fight HIV/AIDS, TB, and Malaria, and Gavi, the Vaccine Alliance. Researchers found that the current financial flow allows donors to provide funds and assistance while maintaining closer control and monitoring throughout the entire project. “We highlight three major trends in global health governance more broadly that relate to this development: towards more discretionary funding and away from core or longer-term funding; towards defined multi-stakeholder governance and away from traditional government-centred representation and decision-making; and towards narrower mandates or problem-focused vertical initiatives and away from broader systemic goals.”
Stories You May Have Missed:
- Federal Hiring Freeze Disrupts USDA’s Food Safety Testing – The transition of the new administration and federal hiring freeze seems to be having some concerning food safety implications. While the FDA has noted that the federal freeze won’t impact the Food Safety Modernization Act, the USDA has highlighted that it is causing issues with the Food Safety Inspection Service (FSIS). “In an internal message sent to FSIS employees on Jan. 18 and obtained by Food Safety News warned that delays in lab tests are expected through at least March 3. The FSIS is responsible for ensuring the safety of meat, poultry, processed egg products and catfish. ‘Effective Jan. 18, 2017, due to a temporary decrease in staffing, results on pathology samples submitted to the FSIS laboratory system will be delayed,’ according to the email sent to all FSIS employees. ‘AMR-01 and rush cases will be given priority status; however turnaround times are expected to be delayed by at least 24 hours on these samples. This is expected to be rectified by March 3, 2017, but is dependent on staffing key vacancies. The Pathology Branch apologies for the inconvenience these delays will cause’.”
- Is Trump Causing a Brain Drain? – Last week’s immigration executive order has many in the science community either unable to travel/return to the U.S. or considering relocation. “The Trump White House’s decision to clamp down on communication from various federal agencies, including the Environmental Protection Agency, has left researchers frightened over political influence seeping into their work. And his executive order has left students and scientists in limbo, removed from their classrooms and work. Advocates are warning that the inhospitable environment will lead, quite quickly, to a brain drain. A young generation of thinkers, academics and researchers might simply look to other countries to conduct their work.”
- The Rise of MCR-1 and the Importance of Understanding the DURC Debate- This week, yours truly is talking to Contagion Live in regards to two very important topics- the rise of antimicrobial resistance and why everyone should understand the DURC debate. It’s easy to get tunnel vision when it comes to science and policy, however so many of these topics are becoming increasingly relevant and in the end, global health security impacts us all.
 GMU Biodefense sent four graduate students to give you a “boots-on-the-ground” viewpoint for the 2017 ASM Biothreats conference. In our special edition post we have a full range of coverage for this three-day conference on biological threats and safety.
GMU Biodefense sent four graduate students to give you a “boots-on-the-ground” viewpoint for the 2017 ASM Biothreats conference. In our special edition post we have a full range of coverage for this three-day conference on biological threats and safety.


 GMU Biodefense Director Talks Growing Threats and Lack of Action
GMU Biodefense Director Talks Growing Threats and Lack of Action