Non-Medical Obstacles Impacting Public Health Responses
GMU Biodefense doctoral alum Jennifer Osetek is discussing non-medical challenges of public health preparedness and responses. “Clearly, for most populations, receiving medical therapies and supplies saves lives; however, medications unable to reach their intended targets prove worthless. ‘Vaccines that remain in the vial are 0% effective’ (Orenstein, Seib, Graham-Rowe, & Berkley, 2014). Outside obstacles stand between patients and the administration of critical health care resources (CHCRs) including medical countermeasures (MCMs), equipment, and supplies. These obstacles can, therefore, result in severe consequences. From a public health perspective, resources that do not reach those affected during an outbreak can mean the difference between a contained disease cluster or the disease spreading and threatening national or even global health security. This is especially relevant as ‘the problem of infectious disease is no longer only one of prevention, but also—and perhaps even more—one of preparedness’ (Lakoff 2008). Obstacles preventing sick people from accessing available resources is not a hypothetical concern any longer. Instead, it is one continually being played out both domestically and internationally in routine and emergency response situations. These obstacles are rooted in various causes and require more specific identification and analysis to prevent inadequate public health responses. Barriers to the delivery of care cost hundreds of thousands of lives simply because they are not defined or incorporated into public health planning and execution operations.”
 Summer Workshop on Pandemics, Bioterrorism, and Global Health Security
Summer Workshop on Pandemics, Bioterrorism, and Global Health Security
Come spend 3.5 days with some of the top minds in the biodefense field this July at our summer workshop. From vaccine development to biosecurity as a wicked problem, we’ll be having frank conversations regarding the toughest issues in health security. Register before May 1st for an early discount and get another discount if you’re a returning student, GMU alum/current student/professor, or registering with a large group.
HHS’ BARDA Funds Its First Marburg Vaccine
In pursuit of making progress against those viruses causing hemorrhagic fevers, HHS has partnered with Public Health Vaccines to help develop a Marburg virus vaccine. “The Biomedical Advanced Research and Development Authority (BARDA), part of the HHS Office of the Assistant Secretary for Preparedness and Response, awarded an initial 2-year, $10 million contract to Public Health Vaccines, LLC to begin development of a vaccine to protect against Marburg infection. ‘This vaccine candidate is the first BARDA has funded against the Marburg virus, and it is an important step toward meeting an urgent public health and biodefense need,’ said BARDA Director Rick Bright, Ph.D. ‘We will leverage our experience in establishing public-private partnerships that bring results that are critical to saving lives and protecting Americans – and possibly people across the globe – from health security threats.’ The Public Health Agency of Canada initially developed the vaccine and licensed it to Public Health Vaccines, LLC. This approach is similar to the one Merck & Co. used to develop its Ebola vaccine. Under the agreement with BARDA, Public Health Vaccines will conduct preclinical development to demonstrate the proof of concept that the vaccine can protect against Marburg virus. If that initial development succeeds, BARDA has the option to provide additional funding for a total of up to $72 million to advance the Marburg virus vaccine through a Phase 2 clinical trial, and begin development of a vaccine candidate against the Sudan ebolavirus, a closely related virus, as well.”
Ebola Outbreak Updates
The outbreak in the DRC has been gaining speed in recent days at eight new cases were reported on Tuesday. “The illnesses lift the overall outbreak total to 968 cases, which includes 903 confirmed and 65 probable infections. Health officials are still investigating 234 suspected cases. Three more people died from Ebola, including two in community settings—one in Katwa and one in Mandima. The other fatality occurred at Butembo’s Ebola treatment center. The developments increase the overall number of deaths to 606. In its weekly diseases and health emergencies update, the WHO’s African regional office said though Katwa health zone is still the main epicenter, responsible for 44% of cases over the past 3 weeks, seven health zones have reported new confirmed cases over the past 3 days and remain a concern. Besides Katwa they include Masereka, Vuhovi, Butembo, Kyondo, Mandima, and Kayina.” Overall, there have been 44 cases reported in the last 5 days, which is deeply concerning and brought an end to the downward trend we were seeing.
WHO Panel Calls for Registry of Human Gene Editing Research
Two days into a panel meeting of gene editing experts and the WHO is calling for a registry to facilitate transparency in human genome editing research. The panel was created as a result of CRISPR baby experiment and is in the process of setting up the registry to help guide work while ensuring safety. “The WHO panel’s statement said any human gene editing work should be done for research only, should not be done in human clinical trials, and should be conducted transparently. ‘It is irresponsible at this time for anyone to proceed with clinical applications of human germline genome editing.’ The WHO’s director-general, Tedros Adhanom Ghebreyesus, welcomed the panel’s initial plans. ‘Gene editing holds incredible promise for health, but it also poses some risks, both ethically and medically,’ he said in a statement. The committee said it aims over the next two years to produce ‘a comprehensive governance framework’ for national, local and international authorities to ensure human genome editing science progresses within agreed ethical boundaries.”
Lab Failures – How Dangerous Pathogens Are Escape Artists
Any research with dangerous pathogens brings with it an inherent risk, whether it be biosafety or biosecurity related. Unfortunately there are a lot of opportunities for failure during such work and recent efforts have looked to incidents to try and identify trends. “It looks like there are many different points of failure — machinery that’s part of the containment process malfunctions; regulations aren’t sufficient or aren’t followed. Human error means live viruses are handled instead of dead ones. Sometimes, these errors could be deadly. ‘If an enhanced novel strain of flu escaped from a laboratory and then went on to cause a pandemic, then causing millions of deaths is a serious risk,’ Marc Lipsitch, a professor of epidemiology at Harvard, told me.” Breaches can occur as a result of carelessness or just pure human error. “The blizzard of dangerous errors over only a few months in 2014, and the additional errors uncovered by subsequent investigations, inspired the US government to change its practices. The government called on all labs that handle secure substances to immediately improve their inventory policies and review their procedures, and to provide written documentation that they’d done so. It launched government-wide reviews to better understand how to safely regulate pandemic pathogens. The FDA began providing better training and conducting periodic audits to make sure that the safety procedures that were ignored in this case are being followed.”
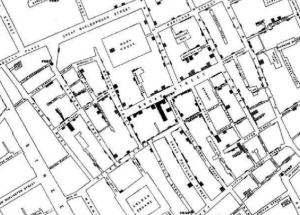 A Modern Take on the Broad St. Pump Outbreak
A Modern Take on the Broad St. Pump Outbreak
GMU Biodefense doctoral student and infectious disease epidemiologist Saskia Popescu is taking a look into an outbreak linked back to contaminated wells and how this reminds us all of the cholera outbreak in 19th century London. “A city, an outbreak, and a contaminated well. Surely this sounds like some kind of modern version of the John Snow cholera outbreak and the Broad St. pump. But unfortunately, it’s this week’s US Centers for Disease Control and Prevention’s Morbidity and Mortality Weekly Report with a much more recent example of how bad sanitation and contaminated water can affect a city. In 2017, a city in Nebraska experienced an outbreak of Campylobacter jejuni that drove home the realities of One Health, the theory that the health of humans, animals, and the environment are all connected. It all began on a March day in 2017, when the Southwest Nebraska Public Health Department got a call regarding a spike in campylobacteriosis cases—5. The condition, campylobacteriosis –infection due to Campylobacter jejuni, is reportable, indicating labs and hospitals are required to report them to the health department. Typically, a single case of Campylobacter was reported in this particular city every 3 years, making the infection quite rare. ”
How A Measles Quarantine Can Lead to Eviction
Vaccine-preventable diseases have been a topic of conversation lately with a surge of measles cases and presence of the anti-vaxxer movement. One particular aspect of public health and managing outbreaks of vaccine-preventable diseases though, is quarantine. A recent article discusses the need for paid medical/sick leave and how those quarantined during outbreaks have faced financial hardships. “The health department persuaded the restaurants where the families worked to not fire them, but the families nevertheless faced steep consequences from avoiding work, according to Archer and Edsall. One family missed so many paychecks that they were evicted. Several people had their phones shut off after unpaid bills racked up. Ultimately, the health department had a collection among its own employees to raise money to donate to the quarantined families. Much of this could have been avoided if the United States had a mandatory-paid-sick-leave policy, Archer and Edsall argue. The Family and Medical Leave Act of 1993 protects the jobs of some workers for up to 12 weeks for medical reasons, but it does not guarantee pay, and it doesn’t cover more than 40 percent of all American workers. Ten states and 33 cities have their own sick-leave policies, but still, 28 percent of American workers lack access to any kind of sick leave. The United States and South Korea are the only countries in the Organization for Economic Cooperation and Development that do not mandate paid sick leave.”
‘TIS the Way to Transport Highly Contagious Patients by Air
“U.S. Air Force personnel conducted training on the Transportation Isolation System (TIS), an enclosure the Defense Department can use to safely transport patients with highly contagious diseases, aboard a C-17 Globemaster III last week in South Carolina. First implemented after the Ebola virus outbreak in 2014, the TIS was engineered to ensure service members get the proper treatment in the event they get infected with any disease during relief missions to affected areas while protecting the aircrew and support personnel. TIS training takes place roughly three times a year and lasts for four days. The training goes from initial donning and doffing protocols for personal protective equipment to actual patient transport and care. This can include treating simulated patients at the “infection scene” all the way to securing them within the TIS unit and even taking part in a simulated in-flight transport.”
Antibiotics, Orchards, and A Citrus Scourge That Instigated Public Health Fear
I’m just going to say it – if I want to know what’s really going on in the world of antimicrobial resistance, I’m looking to Maryn McKenna. Call her our canary in the AMR coal mine. Yet again, she’s giving insight into a concerning avenue for antimicrobial resistance and over usage in the citrus world – this time combatting citrus greening. “The US Environmental Protection Agency (EPA) is in the process of allowing growers to use streptomycin and oxytetracycline as routine treatments, spraying trees several times per year, beginning with the ‘first flush’ of leaves this spring. Growers in the state could end up using as much as 440,000 kilograms of the drugs. Although the compounds, which are both used in human medicine, have been sprayed on other crops in the past and applied in limited amounts to citrus groves, the scale of this application has researchers and public-health advocates alarmed. ‘They are doing a huge experiment with limited monitoring,’ says Steven Roach, a senior analyst in Iowa City at Keep Antibiotics Working, a coalition of research and advocacy groups that has formally objected to the plan with the EPA.” That’s right, as mounting pressure has been focused on reducing antibiotic usage in agriculture, these farmers are planning to use more. Even more frustrating is the lack of evidence for both efficacy of use but also the implications of long-term use. As McKenna notes, “Academic researchers disagree on how much ecological harm or antibiotic resistance will result.” She further points out that “The EPA has specified certain rules to reduce the risk of resistance emerging. Spraying is limited to a few times per year; farm workers must wear full protective clothing; and groves cannot be fertilized with uncomposted manure. It has also set a deadline of seven years for re-evaluating the programme, half the time that it would normally impose for an agricultural chemical.”
Assessing the Need for and Uses of Sequences of Interest Database
You can now find the report on the proceedings from this two-day workshop here. “Over the past decade, the biotechnology economy has experienced remarkable growth, resulting in the rapid expansion of biological knowledge and application. Such advances have lowered the technical and financial barrier to entry for bioexperimentation outside the traditional environments of academia and industry. Together these developments provide exciting new opportunities for scientific growth. However, they create openings for actors with malicious intent to harness readily available tools and techniques to create biological threats or bioweapons. In this report, we present the results of a workshop designed to convene key experts from diverse stakeholder groups to understand how a genetic database of “sequences of interest” (SOIs) can best support stakeholders—government agencies, academic researchers, and commercial groups—to improve the utility, safety, and security of biotechnology research endeavors. The sessions consisted of a mix of presentations, panel discussions, and small and large group discussions. This report should be viewed as an exploratory first step in discussing a very complex topic with broad and often conflicting stakeholder interests.”
Attacks on Medicine Machine Learning: Cyberbiosecurity
“With public and academic attention increasingly focused on the new role of machine learning in the health information economy, an unusual and no-longer-esoteric category of vulnerabilities in machine-learning systems could prove important. These vulnerabilities allow a small, carefully designed change in how inputs are presented to a system to completely alter its output, causing it to confidently arrive at manifestly wrong conclusions. These advanced techniques to subvert otherwise-reliable machine-learning systems—so-called adversarial attacks—have, to date, been of interest primarily to computer science researchers (1). However, the landscape of often-competing interests within health care, and billions of dollars at stake in systems’ outputs, implies considerable problems. We outline motivations that various players in the health care system may have to use adversarial attacks and begin a discussion of what to do about them. Far from discouraging continued innovation with medical machine learning, we call for active engagement of medical, technical, legal, and ethical experts in pursuit of efficient, broadly available, and effective health care that machine learning will enable.”
Stories You May Have Missed:
- H3N2 Cases Keep Flu Activity High – “Though flu activity decreased slightly last week, a wave of H3N2 virus activity has led to severe illnesses across the country and four more children have died from the disease, according to this week’s FluView report from the Centers for Disease Control and Prevention (CDC). This is the second week in a row the CDC recorded more H3N2 cases that H1N1, the virus subtype that dominated the first part of the 2018-19 flu season. ‘H3N2 viruses are typically associated with more severe illness in older adults, and flu vaccine may protect less well against H3N2 illness in older adults, making prompt treatment with flu antivirals in this age group especially important during the current period of H3N2 predominance,’ the CDC said today in a summary of the report.”
- Changing the Game in Pediatric Diagnosis of Serious Bacterial Infections – “Figuring out why that 2-month-old with a fever is crying often includes spinal taps, which are painful and risky, alongside rapid antibiotic treatment to avoid meningitis. Bacterial meningitis can be deadly, especially in infants; the US Centers for Disease Control and Prevention (CDC) reports that between 2003 and 2007, there were 4100 cases of bacterial meningitis reported in pediatric patients in the United States, as well as 500 deaths. Given these rates and the risk of life-threatening infections, it’s not unusual that pediatricians would want to perform a spinal tap or administer antibiotics until further diagnostics can be performed. Fortunately, a new protocol has been developed that could not only make a diagnosis of bacterial infections in infants easier but would remove the need for spinal taps and unnecessary antibiotic treatments. Investigators from the Pediatric Emergency Care Applied Research Network (PECARN) created a new protocol from a study of more than 1800 infants seen across 26 emergency departments in the United States.”







 George Mason Global Health Security Ambassadors Program
George Mason Global Health Security Ambassadors Program






 Gene Drives and Frank Discussions With CRISPR Scientists
Gene Drives and Frank Discussions With CRISPR Scientists






 We’re also excited to announce the following Biodefense MS graduates – Zamawang Almemar, Mariam Awad, Laramie Bradford, Michael Conway, Alexander Dowsett, Sarah Doyle, Stephanie Ellis, Haziq Ghani, Zachary Goble, Stephanie Kiesel, Alexander Rowe, Stephanie Smith, and Alexandra Williams. We’d also like to congratulate three of our biodefense graduate students for their student achievements – Stephanie Smith (Outstanding Biodefense Student), Saskia Popescu (Frances Harbour Award- Biodefense Community Leadership), and Christopher Brown (Outstanding Doctoral Student in Biodefense).
We’re also excited to announce the following Biodefense MS graduates – Zamawang Almemar, Mariam Awad, Laramie Bradford, Michael Conway, Alexander Dowsett, Sarah Doyle, Stephanie Ellis, Haziq Ghani, Zachary Goble, Stephanie Kiesel, Alexander Rowe, Stephanie Smith, and Alexandra Williams. We’d also like to congratulate three of our biodefense graduate students for their student achievements – Stephanie Smith (Outstanding Biodefense Student), Saskia Popescu (Frances Harbour Award- Biodefense Community Leadership), and Christopher Brown (Outstanding Doctoral Student in Biodefense).