Happy Friday fellow biodefense enthusiasts! The Winter Olympics are already starting off with a bang as Korean health officials have confirmed 128 cases of norovirus among security personnel, police officers, and Olympic workers.
WHO Releases List of Blueprint Priority Diseases
The WHO has just released their annual review of the Blueprint list of priority diseases, which includes a special tool for “determining which diseases and pathogens to prioritize for research and development in public health emergency contexts”. While the list includes diseases like Ebola, MERS and SARS, Nipah, and Zika, it is the inclusion of Disease X that highlights the unknowns of infectious diseases in the future. “Disease X represents the knowledge that a serious international epidemic could be caused by a pathogen currently unknown to cause human disease, and so the R&D Blueprint explicitly seeks to enable cross-cutting R&D preparedness that is also relevant for an unknown ‘Disease X’ as far as possible.”
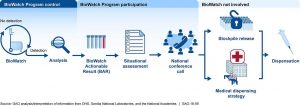
BioWatch Documents Left Behind on Flight
Biowatch officials are doing a bit of a damage control at the moment as multiple copies of an official DHS document regarding performances during a bioterrorism simulation were left behind on an airplane prior to the Super Bowl. “The errant document offered a critique of how officials performed during a simulated release of the infectious disease anthrax in Minneapolis on the day of the Super Bowl. Other sensitive material was also found, CNN said, without further description.” Ironically, it was a CNN employee who discovered the forgotten papers in the seat-back pocket of a commercial plane. “The reports were accompanied by the travel itinerary and boarding pass of the government scientist in charge of BioWatch, the DHS program that conducted the anthrax drills in preparation for Super Bowl LII in Minneapolis. The reports were based on exercises designed to evaluate the ability of public health, law enforcement and emergency management officials to engage in a coordinated response were a biological attack to be carried out in Minneapolis on Super Bowl Sunday.” CNN waited to publish the incident until after Sunday’s game, but the papers revealed significant areas for response improvement during a bioterrorism incident. While not surprising, the identification of such gaps is critical for improvement however, it is the concerning nature of which they were left behind. Vulnerabilities are always present, but that doesn’t mean we want them being aired in the open for nefarious actors to utilize.
 GMU Biodefense PhD Student Selected As ELBI Fellow
GMU Biodefense PhD Student Selected As ELBI Fellow
The Biodefense Graduate Program in the Schar School of Policy and Government at George Mason University is proud to announce that PhD student Yong-Bee Lim has been selected for the prestigious Emerging Leaders in Biosecurity Fellowship (ELBI) program within the Johns Hopkins Center for Health Security. Selected from a group of over 120 international applicants, the 2018 ELBI class includes professionals and scholars with a passion for health security and biosecurity. This year-long fellowship will deepen the biosecurity expertise and broaden the network of those passionate about furthering the field. Yong-Bee was also the recipient of an internship with the Center for Global Security Research at Lawrence Livermore National Laboratory and the SB7.0 Fellowship, which supported his attendance of an international synthetic biology conference in Singapore. His selection also marks the third consecutive year that the Schar School’s Biodefense program has had graduate students named ELBI fellows. Congrats Yong-Bee!
Smallpox Signalling
It’s been a couple of weeks since PLOS One published the horsepox synthesis paper and the truth is that many are still in awe that not only such work was done, but that it was published. Where were broader discussions regarding the merits of this research? Conversations about need to publish it? Moreover, what about the importance of oversight in the private biotech arena and not just those with federal funding? One thing is for certain though, this is a critical moment for life sciences and DURC oversight. “A vaccine against smallpox already exists. The research was carried out unilaterally, without a broader discussion of the merits beforehand. There are apparently few oversight mechanisms when experiments are done with private funding. The restrictions that now cover dual-use research funded by U.S. government — which include an independent review process, and a weighing of the risks and benefits, as well as the ethics — should be expanded to private-sector research.”
Topics of Interest at the DHS Meeting on Sequences of Interest
Synthetic biology has led to the creation of new products, markets, companies, and industries. At the same time, this technology poses potential risks to biosafety and biosecurity, as recently demonstrated by the synthesis of horsepox virus, a cousin of variola, the virus that causes smallpox. On January 29-30, 2018, the Department of Homeland Security (DHS) Science and Technology Directorate sponsored a workshop to discuss the evolving role of databases that contain genetic sequences of pathogens and toxins that pose safety or security concerns, termed “sequences of interest.” The workshop brought together stakeholders from government, industry, and academia to discuss the need for such databases, review current databases and those under development, explore potential applications and users of these types of databases, and consider the potential risks that they pose due to malicious or inadvertent misuse. The workshop provided a valuable opportunity to explore the scientific and technical aspects of constructing such databases, maintenance and sustainability challenges, and the trade-offs involving functionality, accessibility, affordability, confidentiality, and security. While the workshop did not produce a consensus on the best path forward, it played an important role in educating the participants on the most critical issues and facilitating a dialogue among a diverse range of stakeholders on this important topic. The workshop also came at a propitious time as stakeholders grapple with the changing landscape of the biotech industry and advances in DNA synthesis technology. For instance, the International Gene Synthesis Consortium, a group of the leading DNA synthesis companies that have adopted customer and sequence screening protocols to prevent the misuse of their products, has expanded in size and geographic scope and recently updated its biosecurity protocols. The U.S. government is also reviewing the customer and sequence screening guidance it issued in 2010 and is considering whether, and how, to update it. Should a decision be made to proceed, stakeholder engagement would be a significant part of the review, just as it was a significant part of the guidance’s original development.
The US Can’t Afford to Reduce Public Health Funding
GMU biodefense PhD alum Daniel M. Gerstein is evaluating the decision to reduce funding for global epidemic prevention activities and just how dangerous that would be for the United States. “Recent reporting suggests that the Trump administration is preparing to downsize the Centers for Disease Control’s (CDC) global epidemic prevention activities in 39 of 49 countries starting in 2019 when funding first authorized by Congress in 2014 expires. Such a move is potentially dangerous and could place the U.S. at significant risk.” Gerstein points to the notion of shared responsibility in global health security and how important efforts like the GHSA are for global public health. He also notes that the last decade alone has revealed just how undeniable public health is as a national security issue. “Without renewed funding, the long-term outlook could include weakened global disease surveillance and response systems, less capable partner nations and an increased likelihood of global disease outbreaks that would undoubtedly threaten the U.S.” Wanting more information regarding the implications of CDC cutting back on their global health security funding? Check out this article regarding the funding cliff the CDC is about to fall off.
Biosecurity in Putin’s Russia
The latest book from Raymond A. Zilinskas and Philippe Mauger delves into the biodefense world of Putin’s Russia. “In March 2012, at a meeting convened by the recently reelected Russian president Vladimir Putin, Minister of Defense Serdyukov informed Mr. Putin that a plan was being prepared for ‘the development of weapons based on new physical principles: radiation, geophysical wave, genetic, psychophysical, etc.’ Subsequently, in response to concerns expressed both in Russia and abroad, the Russian government deleted the statement from the public transcript of the meeting. But the question remains: Is Russia developing an offensive biological warfare program?”
Global Monitoring of Disease Outbreak Preparedness
The Harvard Global Health Institute has released their new report that is “a step towards developing a shared framework and monitoring mechanism” for outbreak response and preparedness. “This report is primarily intended for the community of policymakers and researchers concerned about the rising risks of domestic, regional, and global infectious disease epidemics, and the collective failure to take the coordinated actions required to reduce such risks. These risks include the expected health, economic, and societal costs that are borne by countries, regions, and even all nations in the case of pandemics (which are worldwide epidemics). These risks also include the consequences of increasing antimicrobial resistance (AMR) and its spread within regions and globally.” The report focuses on strengthening public health capacity as a foundation, improving S&T, reinforcing risk analysis and incentives for action, strengthening global mechanisms, and revised shared monitoring framework and next steps.
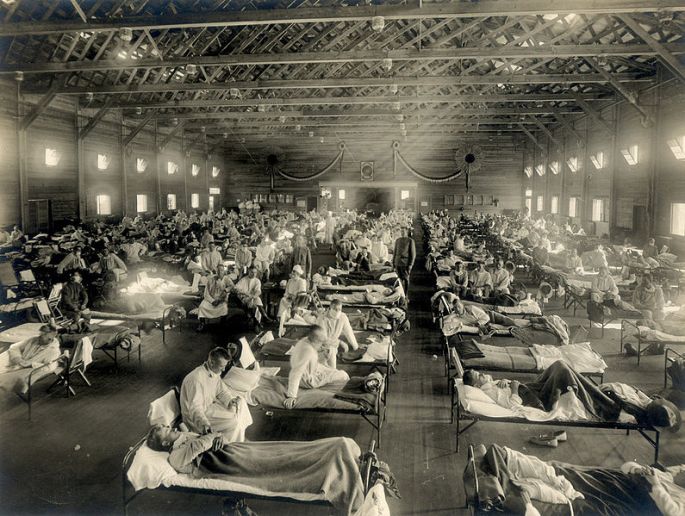
 The Fearsome Future of Flu
The Fearsome Future of Flu
This year has been rough for seasonal flu and while it has been a cold dose of reality in terms of hospital response, are we really learning our lesson or just running from fire to fire? 2018 marks the centennial of the 1918/1919 influenza pandemic and with a larger, more connected population, have we made ourselves safer? The next flu virus with pandemic potential will either enhance our response capabilities or exploit our most damning weaknesses. “And influenza viruses excel at the element of surprise. Few would have guessed Mexico as the origin of that 2009 H1N1 pandemic, for example, notes Fukuda. That outbreak was recognized in San Diego—never considered a hotspot—when a little girl happened to seek treatment at a clinic participating in a study focused on diagnosis, Adalja explains. That’s why he believes it is important to build up the diagnostic capacity for frontline clinicians, and not be satisfied with non-specific diagnoses—failing to pin down the specific microbial cause.” Unfortunately, we are still practicing the methods of frenzied response, delayed information sharing, and dangerous hysteria that can spread even faster through social media. These concerns highlight yet another reason why we need a universal flu vaccine.
Johns Hopkins Center for Health Security Event: Healthcare Disaster Resilience
Don’t miss this event at the National Press Club Holeman Lounge on Thursday, February 22nd from 8:30-10am EST. The Johns Hopkins Center for Health Security will release new policy recommendations for building a more effective disaster health system in the United States. Speakers include Dr. Luciana Borio from the White House National Security Council staff, Sally Phillips of HHS, Linda Langston from the National Association of Counties, as well as Drs.Tom Inglesby, Eric Toner, Monica Schoch-Spana from the Center for Health Security.
 Genetic Sequencing and the Dangers of DIY Genome Editing
Genetic Sequencing and the Dangers of DIY Genome Editing
Nebula Genomics will sequence your genome for just under $1,000 and you can even share it using a blockchain. “Nebula is the brainchild of geneticist George Church, PhD student Dennis Grishin, and graduate Kamal Obbad, all from Harvard. Mirza Cifric, CEO of Veritas Genetics, which offers a genome-sequencing service for $999, is a founding advisor. When you pay to take a DNA test—through 23andMe, Helix, or Ancestry.com, for example—the company that does the testing owns your genetic data. Nebula wants to sequence people’s entire genomes and let them own it, allowing them to earn digital money by sharing it.” While professionals may be providing your genomic data through these companies, many are looking to actually learn to experiment with genes themselves. DIY gene therapy is becoming increasingly accessible, which has many worried and the FDA issuing a warning to the public. The FDA, on “November 21 cautioned against do-it-yourself (DIY) gene therapies. But even as the agency’s statement also warned that selling gene editing kits aimed at homemade therapeutics is illegal, CRISPR–Cas9 kits are available to buy online, and the number of providers is growing, raising concerns that DIY gene therapy enthusiasts could be putting themselves and others at risk.”
Biodefense Policy Landscape Analysis Tool
B-PLAT is a new tool from Pacific Northwest National Laboratory scientists Rachel Bartholomew and Kristin Omberg that aims to help coordinate national biodefense preparedness. It “captures and presents a slew of information about U.S. efforts to protect its citizens and others around the world from threats as diverse as the flu, diseases like Ebola, threats from terrorists, potential risks to water and food supplies, and myriad other concerns. The tool is freely available. In 2017, PNNL chartered an internally funded working group, the Policy Wranglers, to capture relevant biodefense policy directives, public laws, and corresponding sections of the U.S. Code, in a format conducive to visualization. The resulting tool can be utilized to better understand the current state of the U.S. biodefense enterprise.” A presentation on B-LAT will also be made at ASM’s Biothreats next week as part of a town hall on Tuesday.
Stories You May Have Missed:
- US Battles IV Bag Shortage During Record-Setting Severe Flu Season – GMU biodefense PhD student Saskia Popescu is looking at the impacts of this IV bag shortage during the severe flu season. “Even prior to Hurricane Maria, the United States was already battling challenges with meeting the demand for IV fluids; however, the storm crippled Puerto Rico-based manufacturing sites for materials. In a news release on January 16, the FDA reported that they were taking additional steps to combat the shortages, such as ‘asking companies to submit data to extend expiration dates for these products.’ They noted that, ‘if expiration dates can be safety extended [for these products], it would allow some near-expiry product that remains at the hospital level to be used’.”
- DARPA Program to Track Genetic Markers for Past WMD Material Exposure – “The program is being called Epigenetic Characterization and Observation (ECHO), and its endgame is the creation of a field-deployable system that could analyze someone’s epigenome and identify markers of whether or not–in that person’s entire lifetime–been exposed to WMD-associated materials. DARPA officials are billing it as worlds quicker than sending to a lab to test for biological or chemical agents in clothing or hair.”
Thank you for reading the Pandora Report. If you would like to share any biodefense news, events, or stories, please contact our Editor Saskia Popescu (biodefense@gmu.edu) or via Twitter: @PandoraReport